 Cadila Healthcare – the 4th largest player in the Indian pharma market with 16 of the group’s brands feature amongst the top 300 pharma brands in India as per AWACS March 2016, has issued a voluntary recall of 14,808 bottles of Bromocriptine Mesylate Capsules of 5 mg 30-count bottles.
Cadila Healthcare – the 4th largest player in the Indian pharma market with 16 of the group’s brands feature amongst the top 300 pharma brands in India as per AWACS March 2016, has issued a voluntary recall of 14,808 bottles of Bromocriptine Mesylate Capsules of 5 mg 30-count bottles.
Bromocriptine Mesylate is a prescription drug that is approved by the FDA (USA) for use in the treatment, prevention, or management of the certain medical conditions and diseases, including:
- Hyperprolactinemia-Associated Dysfunctions
- Acromegaly
- Parkinson’s Disease
The product was manufactured at Cadila Healthcare, Ahmedabad and Distributed by Zydus Pharmaceuticals, USA. Cadila was slapped by the US FDA Warning Letter with a warning letter for Moraiya formulation facility and Ahmedabad API facility (Zyfine) earlier during the year. This recall has been initiated on the same grounds.
The recall was already initiated from the 9-May but was reported to the FDA only on the 2-Sep. Most of the products recalled are from a single batch of 2016 expiry and 3 batches of 2017 expiry.
Why the Big Deal?
While not all details have been divulged by the company or the FDA, the simple reason stated is “Failed impurities/degradation specifications: Out of specification results noticed in related substance test during analysis of 24 months long term (25 degree Celsius /65% RH) stability samples of two batches.”
The purpose of stability testing is to provide evidence on how the quality of a drug substance or drug product varies with time under the influence of a variety of environmental factors, such as temperature, humidity, and light, and to establish a retest period for the drug substance or a shelf life for the drug product and recommended storage conditions.
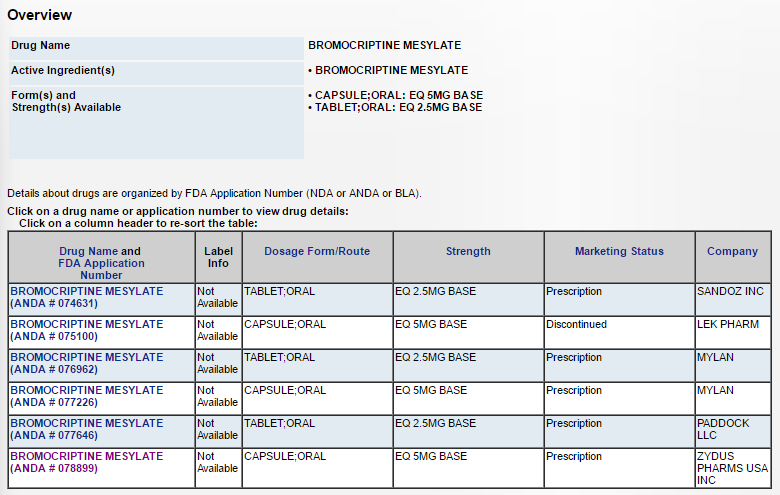
Currently other than Zydus, Mylan and Lek Pharma has approvals for this product.
What is a Class II recall?
The recall has been initiated only in the United States for now, but there is potential for recall from other countries if the same product under the same batch has been sold across other nations. The call has been classified as a Class II recall.
This happens in a situation in which use of or exposure to a violative product may cause temporary or medically reversible adverse health consequences or where the probability of serious adverse health consequences is remote.
This paves the way for any future potential law suits, thereby adding even more stress on the revenues from this product. The recall status is “Ongoing” which means that currently there is no end in sight on the recall of this product.
What now?
This is not the first time that Cadila is recalling products from its Ahmedabad based manufacturing unit. There are already over 4 recalls initiated from this unit just in 2016. Let’s see whether the stock takes a beating today or have the markets already factored in this recall.
Holdings: Analyst and family do own stock listed above.
![]()
Disclaimer
Nothing in this newsletter is financial advice and should not be construed as such. Please do not take trading decisions based solely on the matter above; if you do, it is entirely at your own risk without any liability to Capital Mind. This is educational or informational matter only, and is provided as an opinion.
Disclosure: The authors at Capital Mind have positions in the market and some of them may support or contradict the material given above, or may involve a direction derived from independent analysis.



