
European Medical Agency has accepted the submission for the review of the biosimilar Pegfilgrastim co-developed by Mylan & Biocon.
The proposed biosimilar Pegfilgrastim is one of the six biologic products co-developed by Mylan and Biocon for the global marketplace.
The company in its press release mentioned that not only does the application include clinical data from pivotal Pharmacokinetic / Pharmacodynamic (PK / PD) and confirmatory efficacy, safety and immunogenicity studies completed earlier in 2016, but it also includes analytical, functional and pre-clinical data.
Both the companies hope to present the results from the studies at the prestigious European Society of Medical Oncology (ESMO) Annual Congress to be held in Copenhagen in October, 2016.
While Mylan will own the exclusive commercialization rights for the U.S., Canada, Japan, Australia, New Zealand, EU and European Free Trade Association countries, Biocon has co-exclusive commercialization rights with Mylan for the product in the rest of the world.
Once approved, this product will enable enhanced access to a cost-effective alternative for patients undergoing chemotherapy in the EU as per Arun Chandavarkar, CEO and Joint Managing Director, Biocon.
Mylan President Rajiv Malik said that “This milestone in our Pegfilgrastim program represents yet another important step in bringing more affordable versions of these critical products to market, with Europe representing an exciting opportunity for Mylan in this area.”
While the news hit the markets post trading hours, Biocon closed in red at Rs. 700.60/- down by 1.81% and Mylan was trading -1.23% down.
What is Pegfilgrastim?
Pegfilgrastim is prescribed for cancer patients to help them with some of the side-effects of their treatment. It reduces the duration of neutropenia (low levels of neutrophils, a type of white blood cell that fights infections) and the incidence of febrile neutropenia (neutropenia with fever) that are a result of their chemotherapy treatment.
Biocon and Mylan are exclusive partners on a broad portfolio of biosimilars and generic insulin analogs. The proposed biosimilar Pegfilgrastim is one of the six biologic products co-developed by Mylan and Biocon for the global marketplace. Mylan has exclusive commercialization rights for the proposed biosimilar Pegfilgrastim in the U.S., Canada, Japan, Australia, New Zealand and in the European Union and European Free Trade Association countries. Biocon has co-exclusive commercialization rights with Mylan for the product in the rest of the world.
You might also be interested in Mylan gets ‘all clear’ from EU regulators for $7.2 billion Meda deal
About the Partners
Global pharmaceutical major Mylan offers a growing portfolio of more than 1,400 generic and branded pharmaceuticals, including antiretroviral therapies on which approximately 50% of people being treated for HIV/AIDS in the developing world depend. Mylan’s global R&D and manufacturing platform includes more than 50 facilities.
India’s largest and fully-integrated, innovation-led biopharmaceutical company Biocon is an an emerging global biopharmaceutical enterprise serving customers in over 100 countries.
Biocon has successfully developed and taken a range of Novel Biologics, Biosimilars, differentiated Small Molecules and affordable Recombinant Human Insulin and Analogs from ‘Lab to Market’. It has a rich pipeline of Biosimilars and Novel Biologics at various stages of development including Insulin Tregopil, a high potential oral insulin analog.
The company as well announced its quarterly results for the period ended June, 2016. This was again post market hours and here are some highlights: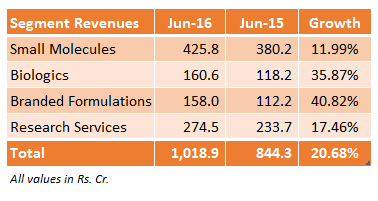
- Revenue from operations increased from Rs. 813 crore to Rs. 982 crore.
- Total Comprehensive Income increased by Rs. 30.9 crore to Rs. 150.5 crore
![]()
Disclaimer
Nothing in this newsletter is financial advice and should not be construed as such. Please do not take trading decisions based solely on the matter above; if you do, it is entirely at your own risk without any liability to Capital Mind. This is educational or informational matter only, and is provided as an opinion.
Disclosure: The authors at Capital Mind have positions in the market and some of them may support or contradict the material given above, or may involve a direction derived from independent analysis.




