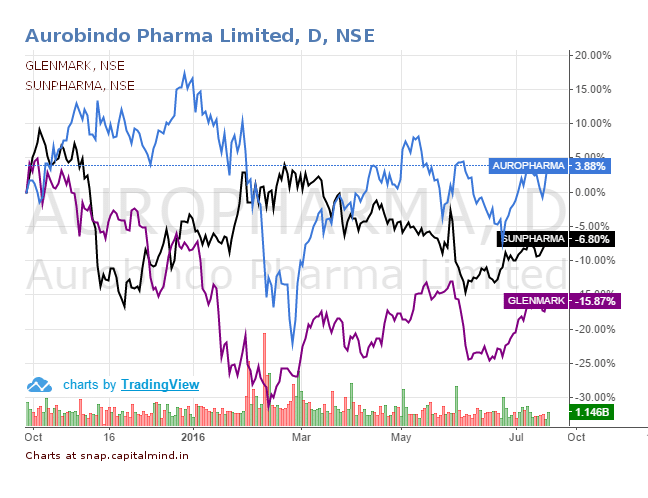
This does not happen everyday but today, the US FDA happened to approve the ANDA filings of 3 pharmacos (Aurobindo Pharma, Glenmark Pharmaceutical and Sun Pharmaceuticals) for the bio-equivalence CRESTOR drug manufactured by IPR Pharmaceuticals Inc.
Though all 3 pharmacos have received the ANDA approval, Aurobindo Pharma was the generics manufacturer (Generic drugs approved by the FDA have the same quality and strength as brand-name drugs. Generic drug manufacturing and packaging sites must pass the same quality standards as those of brand-name drugs) eligible for the 180 days exclusivity. This was because Aurobindo was one of the first ANDA applicants to submit a substantially complete ANDA with a paragraph IV certification.
A Paragraph IV filing in the United States is made when an ANDA applicant believes its product or the use of its product does not infringe on patents or where the applicant believes that such patents are not valid or enforceable. Paragraph IV basically means cases where the patent has not been infringed upon under the Hatch-Waxman Act.
As per IMS, this approved product has an estimated market size of US$ 6.7 billion for the twelve months ending May 2016.
Watson Pharmaceuticals Inc. of Parsippany, New Jersey was the first company to receive the approval to market generic rosuvastatin calcium. A settlement in a patent infringement suit gave the Watson the sole rights to start selling its version in early May, 67 days before the exclusivity ended on 8 July.
More about Rosuvastatin Calcium Tablets:
It is an antihyperlipidemic to prevent cardiovascular disease. It is used to reduce elevated total-C, LDL-C, ApoB, non HDL-C, and TG levels and to increase HDL-C in patients with primary hypercholesterolemia and mixed dyslipidemia.
The approval received is for 5 mg (base), 10 mg (base), 20 mg (base) and 40 mg (base) versions.
Disclosure: Analyst owns the above stocks. Please assume we are biased.
![]()
Disclaimer
Nothing in this newsletter is financial advice and should not be construed as such. Please do not take trading decisions based solely on the matter above; if you do, it is entirely at your own risk without any liability to Capital Mind. This is educational or informational matter only, and is provided as an opinion.
Disclosure: The authors at Capital Mind have positions in the market and some of them may support or contradict the material given above, or may involve a direction derived from independent analysis.



