
Unichem Laboratories is a specialty pharmaceutical company with a large basket of branded generics in India and several other markets across the world. (latest investor presentation here)
With formulations forming the core of Unichem’s business, the company also manufactures active pharmaceutical ingredients (APIs or bulk actives). In addition, it has several pharma products that address relevant and growing therapeutic areas like gastroenterology, cardiology, diabetology, psychiatry, neurology, anti-bacterials, anti-infectives and pain management among others.
Unichem is headquartered in Mumbai, India, and has six drug manufacturing locations across the country with several of the company’s facilities being accredited by US FDA, ISO, UK MHRA (earlier MCA), MCC (South Africa), WHO (Geneva) and TGA (Australia).

The company announced that it has received ANDA approval from the United States Food and Drug Administration (USFDA) for Donepezil Hydrochloride Tablets USP.
Donepezil Hydrochloride Tablets USP 5mg and 10mg are therapeutically equivalent to Aricept Tablets, 5mg and 10mg of Eisai, Inc. (USA).
Donepezil Hydrochloride is a reversible inhibitor of the enzyme acetylcholinesterase which is used to improve cognition and behavior of people with Alzheimer’s, but does not slow the progression of or cure the disease. The product will be commercialized from Unichem’s Goa plant. Active Pharmaceutical Ingredient will also be made in house at Roha API Plant.
Unichem Laboratories Limited now has total of 21 molecules approved from USFDA.
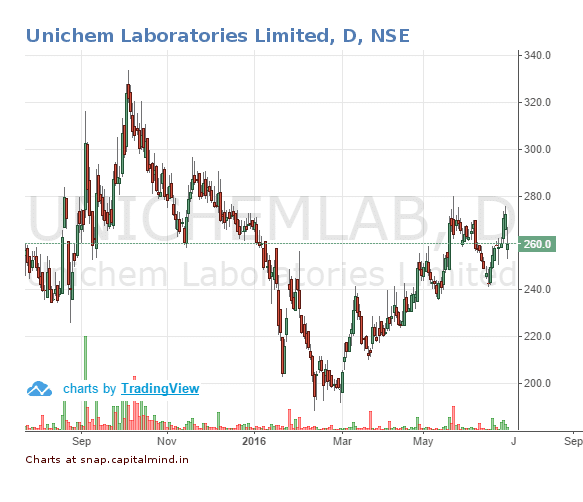
![]()
Disclaimer
Nothing in this newsletter is financial advice and should not be construed as such. Please do not take trading decisions based solely on the matter above; if you do, it is entirely at your own risk without any liability to Capital Mind. This is educational or informational matter only, and is provided as an opinion.
Disclosure: The authors at Capital Mind have positions in the market and some of them may support or contradict the material given above, or may involve a direction derived from independent analysis.



